
Higher the pH value, stronger will be the base. Acids have a pH lesser than 7.0 and the lower it is, the stronger the acid becomes. They both conduct electricity depending on the dissociation of ions. They can react with bases to produce salts and water. Acids taste sour and create a stinging feeling on the mucous membranes. Sodium Hydroxide (NAOH) and Ammonia (NH3)īases have a slippery feel on fingers and taste bitter. For example, NaOH (Sodium Hydroxide).Īcetic acid i.e.CH3COOH and Sulfuric acid There is one exception to his rule, CH3COOH = Acetic Acid (vinegar)Ī base has a chemical formula with OH at the end of it. Taste bitter.ĭepends on concentration of the hydronium ionsĭepends on concentration of the hydroxide ionsĮlectrolytes, conduct electricity (because electrolytes), react with many metals.Įlectrolytes, conduct electricity, ranges from insoluble to so soluble that they can react with water vapor.Īcids free hydrogen ions (H+) when mixed with water.īases free hydroxide ions (OH-) when mixed with water.Īn acid has a chemical formula with H at the beginning of it. Frequently solids except ammonia which is a gas. Taste sour.īases feel slippery because of the reaction of the base with the oils of your hand. Greater than 7.0 and could go up to 14 in case of stronger bases.ĭepending on the temperature, acids can occur in solid, liquid or gaseous form. PH (measure of concentration of hydrogen ions in a solution) Bronstead Lowry Definition: A base is any substance which accepts a proton. Bronstead Lowry Definition: An acid is an substance which donates a proton.Īrrhenius Definition: A base is an aqueous substance that can accept hydrogen ions. The Which OSHA Standards Apply webpage can help identify other standards that may be applicable to your worksite.Comparison chart Acid versus Base comparison chartĪrrhenius Definition: An acid is any chemical compound which when dissolved in water gives a solution with a hydrogen ion activity greater than in pure water. 1450 - occupational exposure to hazardous chemicals in laboratories Subpart Z - toxic and hazardous substances Subpart J - general environmental controls Subpart I – personal protective equipment Subpart G - occupational health and environmental control Note: Please also check the standards information and activity webpage to see if there has been any recent or upcoming regulatory activity on this topic. OSH has adopted the following standards which are applicable to acids and bases in North Carolina. The consultative services bureau provides free and confidential onsite consultation regarding worksite safety and health hazards. More related information can be found on the following safety and health topic pages: hazard communication, personal protective equipment, respiratory protection and medical services and first aid. Lastly, the NCDOL Library offers free safety and health videos (including streaming video services) and related research assistance on consensus standards (i.e., ANSI, NFPA, NEC).Īn hazard communication program, hazardous chemical program, and personal protective equipment program are available for employers to download and adapt to their specific conditions. IIn addition, the education, training and technical assistance bureau provides free online safety and health training and outreach services (i.e., speaker's bureau requests, safety booths) upon request. Each of these presentations should be modified to address site-specific conditions and hazards. 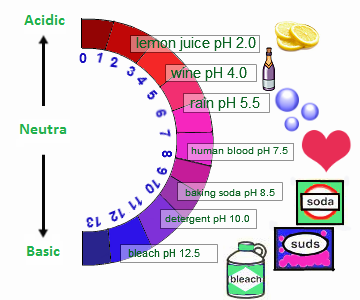
The presentations on hazard communication and personal protective equipment are available to assist employers in training their staff. What resources are available to assist employers? Failure to do so can result in death due to pulmonary edema or heart disorders. 
Persons exposed to hydrogen fluoride should seek prompt immediate medical attention. This prevents the formation and accumulation of shock-sensitive perchlorates. Work with perchloric acid must always be done in dedicated, specially-designed fume hoods that are equipped with wash down capability of the exhaust ductwork. Employers must provide employees with the necessary hazard information and appropriate personal protective equipment, especially eye and face protection and impervious gloves. What can I do to protect myself and others?Įmployees using strong acids and bases must understand the hazards of each of the chemicals they use.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
